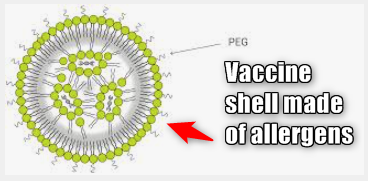
Official. Majority of Adverse Reactions are Due to Allergic Reaction to Lipid Nano-particle Ingredients
Are you allergic to anything?
The rushed mRNA serum was the very first time the toxic chemical "PEG" (Polyethylene glycol) has been used in a vaccine, but it was never tested on people who had allergic reactions to the compounds, so how could it ever be deemed as safe? (Reference 1)
Many people are allergic to PEG and it can have a very severe reaction when a susceptible person is exposed to it. It is commonly found in hair dye so if you have a reaction to hair dye, you most likely have a reaction to the mRNA drug.
It would look like this:
Were we told about this in New Zealand?
- Be of acceptable quality.
- Be fit for a particular purpose that you asked about.
- Match the description given in advertisements or sales brochures, or by the sales assistant.
I now know that a crime has been committed
MEDSAFE QUOTE: "Individuals should not receive the Comirnaty vaccine if they have a history of anaphylaxis to any of the ingredients in the vaccine"
In Summary
- The mRNA Serum contains antigens and was a known risk
- It is highly likely that these allergens cause most if not all adverse reactions
- This factor has not been made obviously aware to the public so that they could make an informed choice
- The crime of coercion has occurred at the highest levels
Conclusion
Now that we know what is causing most if not all of the adverse reactions, people can now be treated for the conditions correctly. Spread the word.
My family is vaccine injured. I have been working on a Hypothesis and a protocol to deal with the inflammatory condition that most vaxxed people appear to have. A swelling of the lymphatic system and possible leaky lymphatics.
End of report.
References:
(Reference 2)
Publisher: Allergy, Asthma & Clinical Immunology (AACI), the official journal of the Canadian Society of Allergy and Clinical Immunology (CSACI)
Topic: Polyethylene glycol as a cause of anaphylaxis
Reference: https://aacijournal.biomedcentral.com/articles/10.1186/s13223-016-0172-7
Published Date: 13 December 2016
Authors: Katharina Wylon, Sabine Dölle & Margitta Worm
Abstract:
Background
Polyethylene glycols (PEGs) or macrogols are polyether compounds and are widely used as additives in pharmaceuticals, cosmetics, and food.
Case report
We report on a Caucasian patient experiencing recurrent severe allergic reactions to several drugs. An extensive diagnostic workup including skin prick tests, intradermal tests (IDT) and a double-blind oral challenge was performed to identify the trigger of anaphylaxis. In the present case hypersensitivity to the additive polyethylene glycol was confirmed by an IDT suggesting an Immunoglobulin E-dependent mechanism as a cause of the reaction.
Conclusion
Potential life-threatening hypersensitivity reactions to hidden molecules like macrogol may be under diagnosed. Cases of immediate-type PEG hypersensitivity were reported with increasing frequency. The awareness regarding the allergenic potential of PEG should be raised and a proper product labeling is crucial to prevent PEG mediated hypersensitivity.
(Reference 3)
Publisher: Science.org
Topic: Suspicions grow that nanoparticles in Pfizer's COVID-19 vaccine trigger rare allergic reactions. Life-threatening responses seen in at least eight people could be linked to polyethylene glycol, known to trigger reactions to some drugs
Published Date: 21 December 2020
Authors: JOP DE VRIEZE
Abstract
(1) Severe allergy-like reactions in at least eight people who received the COVID-19 vaccine produced by Pfizer and BioNTech over the past 2 weeks may be due to a compound in the packaging of the messenger RNA (mRNA) that forms the vaccine's main ingredient, scientists say. A similar mRNA vaccine developed by Moderna, which was authorized for emergency use in the United States on Friday, also contains the compound, polyethylene glycol (PEG).
(2) PEG has never been used before in an approved vaccine, but it is found in many drugs that have occasionally triggered anaphylaxis—a potentially life-threatening reaction that can cause rashes, a plummeting blood pressure, shortness of breath, and a fast heartbeat. Some allergists and immunologists believe a small number of people previously exposed to PEG may have high levels of antibodies against PEG, putting them at risk of an anaphylactic reaction to the vaccine.
(Reference 4)
Publisher: IDSA and OXFORD Academic
Topic: Intravenous Injection of Coronavirus Disease 2019 (COVID-19) mRNA Vaccine Can Induce Acute Myopericarditis in Mouse Model
Reference: https://academic.oup.com/cid/article/74/11/1933/6353927
Published Date: 18 August 2021
Authors: Can Li, Yanxia Chen, Yan Zhao, David Christopher Lung, Zhanhong Ye, Wenchen Song, Fei-Fei Liu, Jian-Piao Cai, Wan-Man Wong, Cyril Chik-Yan Yip, Jasper Fuk-Woo Chan, Kelvin Kai-Wang To, Siddharth Sridhar, Ivan Fan-Ngai Hung, Hin Chu, Kin-Hang Kok, Dong-Yan Jin, Anna Jinxia Zhang, Kwok-Yung Yuen
Abstract
Background
Post-vaccination myopericarditis is reported after immunization with coronavirus disease 2019 (COVID-19) messenger RNA (mRNA) vaccines. The effect of inadvertent intravenous injection of this vaccine on the heart is unknown.
Methods
We compared the clinical manifestations, histopathological changes, tissue mRNA expression, and serum levels of cytokine/chemokine and troponin in Balb/c mice at different time points after intravenous (IV) or intramuscular (IM) vaccine injection with normal saline (NS) control.
Results
Although significant weight loss and higher serum cytokine/chemokine levels were found in IM group at 1–2 days post-injection (dpi), only IV group developed histopathological changes of myopericarditis as evidenced by cardiomyocyte degeneration, apoptosis, and necrosis with adjacent inflammatory cell infiltration and calcific deposits on visceral pericardium, although evidence of coronary artery or other cardiac pathologies was absent. Serum troponin level was significantly higher in IV group. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike antigen expression by immunostaining was occasionally found in infiltrating immune cells of the heart or injection site, in cardiomyocytes and intracardiac vascular endothelial cells, but not skeletal myocytes. The histological changes of myopericarditis after the first IV-priming dose persisted for 2 weeks and were markedly aggravated by a second IM- or IV-booster dose. Cardiac tissue mRNA expression of interleukin (IL)-1β, interferon (IFN)-β, IL-6, and tumor necrosis factor (TNF)-α increased significantly from 1 dpi to 2 dpi in the IV group but not the IM group, compatible with presence of myopericarditis in the IV group. Ballooning degeneration of hepatocytes was consistently found in the IV group. All other organs appeared normal.
Conclusions
This study provided in vivo evidence that inadvertent intravenous injection of COVID-19 mRNA vaccines may induce myopericarditis. Brief withdrawal of syringe plunger to exclude blood aspiration may be one possible way to reduce such risk.
(Reference 5)
Publisher: CNN
Topic: Pfizer's Covid-19 vaccine study excluded people with a history of severe allergic reactions, data shows
Published Date: 10 December 2020
Authors: Ben Westcott, Adam Renton, Eoin McSweeney, Nada Bashir, Mike Hayes, Meg Wagner and Melissa Macaya, CNN
Abstract
The company excluded people with a history of severe allergic reactions to vaccines from their SARS-COV-2 RNA vaccine trial, according to Pfizer data released by the US Food and Drug Administration on Tuesday.
Participants with a “history of severe adverse reaction associated with a vaccine and/or severe allergic reaction (eg, anaphylaxis) to any component of the study intervention(s)” were not included in the pool of 44,000 trial volunteers.
According to the US Food and Drug Administration's evaluation of the Pfizer data, there was a slight increase in events that may be considered allergic reactions in the vaccine group. The FDA’s independent analysis said “A slight numerical imbalance of adverse events potentially representing allergic reactions, with more participants reporting hypersensitivity-related adverse events in the vaccine group (137 [0.63%]) compared with the placebo group (111 [0.51%])”
(Reference 6)
Publisher: NZ MOH
(1) 'If you have had previous reactions to vaccines or injections' does not make it clear enough. The question should be 'If you have allergic reactions' as a person may not remember the results of previous childhood reactions and the statement does not include PEG reactions which is the actual issue because PEG has never been in a vaccine before and is known to be the cause of reaction and not previous vaccines.
(2) The actual information of importance is not known by the trained government administration team, neither is it explained on the brochures or consent form, so how would anyone know?
(3) For many, it was not their choice. They were forced into the decision by the illegal activity of coercion (secret or illegal cooperation or conspiracy in order to deceive others). That is a crime:
(Reference 7)
Publisher: New Zealand Medicines and Medical Devices Safety Authority (MEDSAFE)
Topic: Anaphylaxis following vaccination: focus on Comirnaty
Published Date: 3 June 2021
Authors: MEDSAFE
Abstract
Risk factors for anaphylaxis to Comirnaty vaccine
Individuals should not receive the Comirnaty vaccine if they have a history of anaphylaxis to any of the ingredients in the vaccine.1,9,10 The vaccine ingredients are listed in the New Zealand data sheet (see section 2 for the active ingredient and section 6.1 for the list of excipients).9
Polyethylene glycol (PEG), also known as macrogol, is an ingredient in mRNA COVID-19 vaccines. PEG is present in many different types of medicines and is recognised as an allergen that can trigger anaphylaxis in some people.1,10–13
Individuals with a history of an anaphylaxis-type reaction to any other substance have an increased risk of an anaphylactic response to mRNA COVID-19 vaccines. (These individuals can still receive the vaccine but should be observed for a minimum of 30 minutes and be given clear advice on symptoms of anaphylaxis and how to call for help, before leaving the vaccination facility.)
Treatment of anaphylaxis
Adrenaline is the first-line treatment for anaphylaxis and should be given without delay as soon as anaphylaxis is suspected.6,14,15
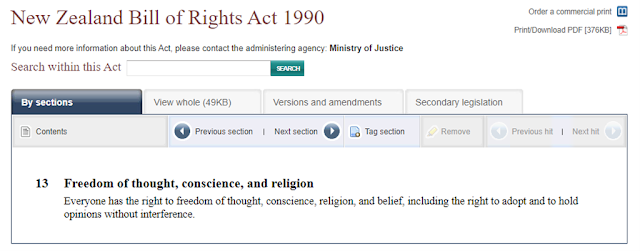
REF: https://www.legislation.govt.nz/act/public/1990/0109/latest/DLM225512.html








Comments
Post a Comment